|
1/2/2024 0 Comments Element table groups 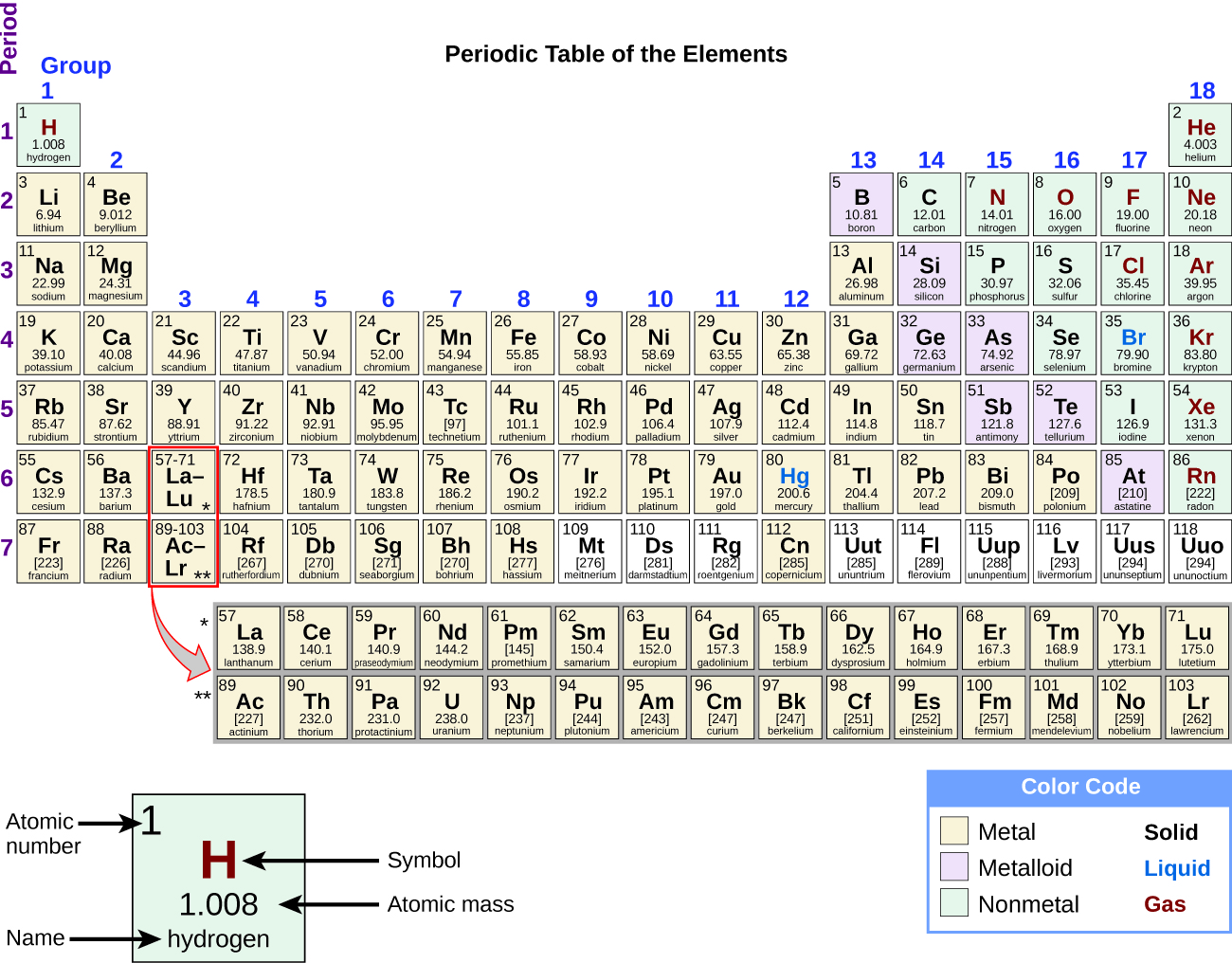
In the Periodic Table, the elements are arranged in order of increasing atomic number. Given the outermost energy level electron configuration for an element, determine its family on the Periodic Table.Īlkali Metals Have One Electron in Their Outer Energy Level.Identify the columns in the Periodic Table that contain 1) the alkali metals, 2) the alkaline earth metals, 3) the halogens, and 4) the noble gases, and describe the differences between each family's electron configuration.Describe the patterns that exist in the electron configurations for the main group elements. 
We will use it for the next two chapters. We will need to remember the sub-level filling groups in the Periodic Table. In this lesson, we will focus on the connection between the electron configuration and the main group elements of the Periodic Table. The orbital representation was learned in an earlier chapter but like many of the skills you learn in chemistry, it will be used a great deal in this chapter and in several chapters later in the course. The orbital representation method for representing electron configuration is shown below. An understanding of these electron configurations will prove to be invaluable as we look at bonding and chemical reactions. With the introduction of electron configurations, we began to get a deeper understanding of the Periodic Table. 7 The Nitrogen Family Has 5 Electrons in the Outer Energy Level.6 The Oxygen Family Has 6 Electrons in the Outer Energy Level.5 Halogens Have 7 Electrons in Their Outer Energy Level.4 Noble Gases Have 8 Electrons in Their Outer Energy Level.3 Alkaline Earth Elements Have Two Electrons in Their Outer Energy Level.2 Alkali Metals Have One Electron in Their Outer Energy Level. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
